When One Catalytic Cycle Is Not Enough
The installation of alkyl substituents through C(sp³)-C(sp³) coupling is in high demand in synthetic and medicinal chemistry. While some strategies towards this goal employ Ni-catalysed cross couplings, this transformation remained challenging due to the difficulty of obtaining cross-selectivity between fleeting aliphatic radicals and alkyl electrophiles.
Recently published research in JACS followed a two-stage evolution from a “single Ni catalyst” approach to a “dual Ni catalyst” approach. Through refinement of mechanistic hypotheses and reaction conditions, the authors identified a potential divergence of the Ni precatalyst to two active species with orthogonal roles. This led to a shift in optimization efforts toward strategically designing a dual Ni catalyst system that ultimately enabled significant reactivity enhancement.
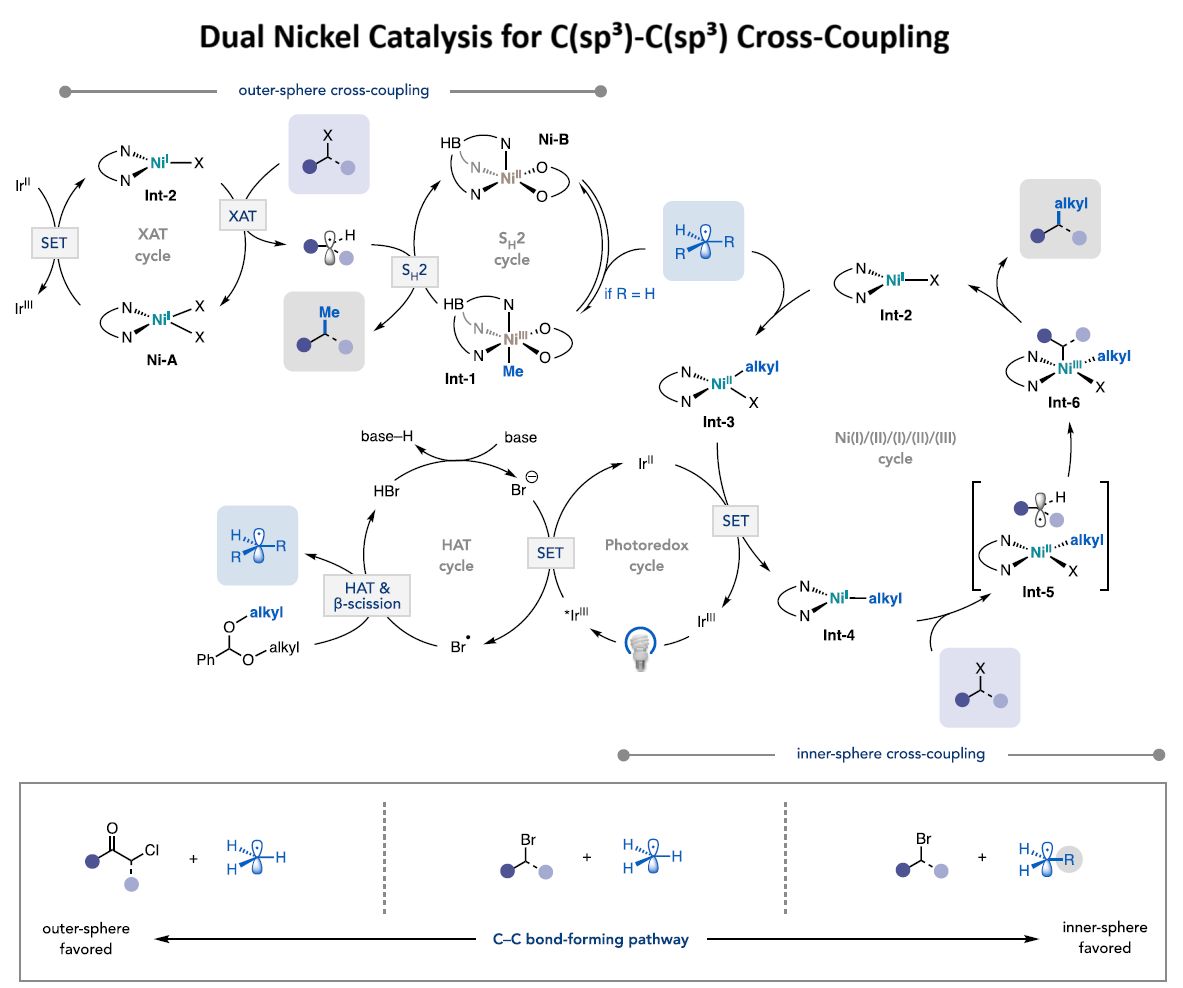
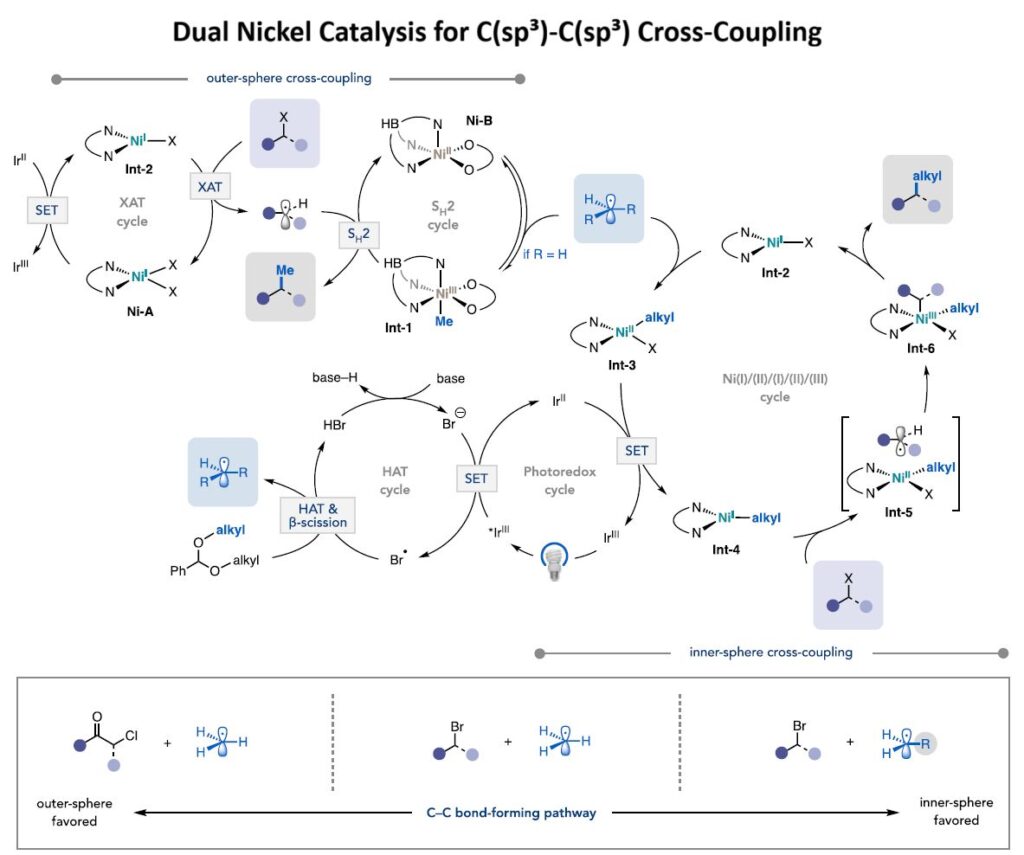
How it works (see figure below)
1) Blue light activates an iridium photocatalyst to generate methyl radicals from benzaldehyde dimethyl acetal (or alkyl radicals from dialkyl acetal) through hydrogen atom transfer and β-scission.
2) This methyl (or alkyl) radical then participates in two concurrent coupling pathways:
𝐎𝐮𝐭𝐞𝐫-𝐬𝐩𝐡𝐞𝐫𝐞 𝐩𝐚𝐭𝐡𝐰𝐚𝐲: Upon adding to Ni-B, methyl (or alkyl) radical can undergo outer-sphere coupling (i.e., S_H2) with a halide-derived alkyl radical generated via Ni-mediated XAT.
𝐈𝐧𝐧𝐞𝐫-𝐬𝐩𝐡𝐞𝐫𝐞 𝐩𝐚𝐭𝐡𝐰𝐚𝐲: The acetal-derived radical adds to (bpy)Ni^I(Br) to give (bpy)Ni^II(alkyl)(Br). Reduction via Ir^II-catalyzed SET furnishes a (bpy)Ni^I(alkyl) species poised for activating the alkyl halide via XAT. Following halide activation, C-C bond formation occurs via radical rebound onto Ni, followed by inner-sphere reductive elimination from the resulting Ni^III intermediate to forge the alkyl-alkyl coupled product and regenerate (bpy)Ni^I(Br).
The two Ni catalysts work cooperatively to overcome the selectivity challenges inherent in using highly reactive alkyl radicals with alkyl electrophiles.
My highlights
➔ Primary focus was on methylation but alkylation with diverse substrates has been also demonstrated
➔ Efficient C(sp³)-C(sp³) coupling across a range of primary and secondary chlorides and bromides
➔ Delivers high yields (up to 80%)
➔ Two orthogonal Ni catalysts outperformed a traditional single-catalyst systems
➔ Successful late-stage functionalization on complex molecules, including those derived from celecoxib and lenalidomide
➔ Preserves stereochemistry in enantioenriched substrates
This dual nickel catalysis strategy represents the first application of cooperative identical transition metal catalysis to alkyl-alkyl cross-coupling.
Share your thoughts
Will dual catalysis with single metal gain importance in modern synthetic chemistry?
Leave your comment here.
Full manuscript: https://doi.org/10.1021/jacs.5c10906