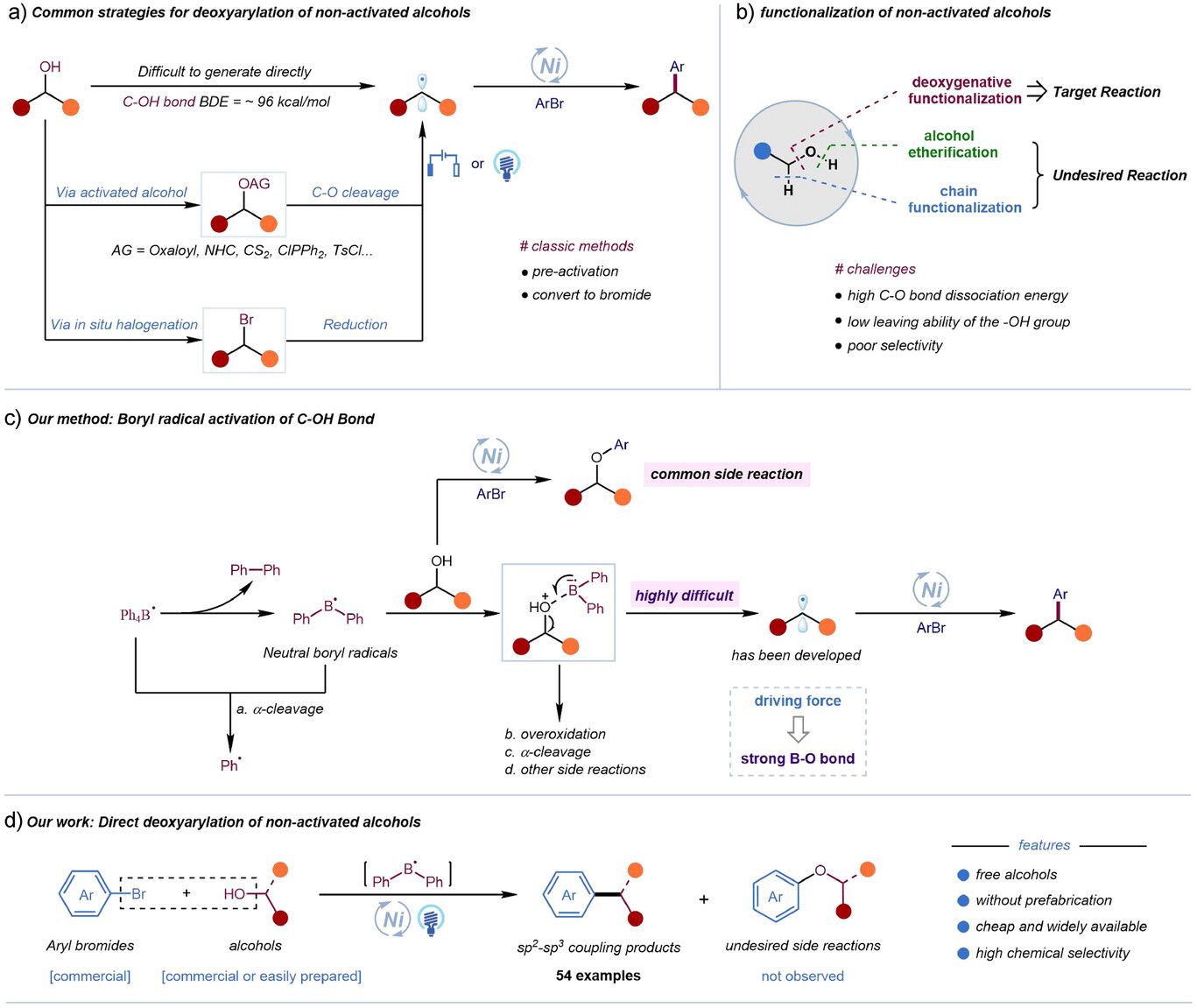
Alcohols as Building Blocks for C-C Cross Coupling
Until now, direct activation of the C─O bond was an unresolved problem, mainly due to high dissociation energy (approximately 96 kcal/mol) and the limited dissociation capacity of the hydroxyl group.
A new photoredox methodology published in Angewandte Chemie tackled this challenge by developing a direct deoxygenative cross-coupling of non-activated primary and secondary alcohols with aryl bromides, no pre-functionalization required.
Core innovation
The use of neutral diphenylboron radicals as hydroxyl activation agents for direct C-O bond activation that enables photocatalytic deoxyarylation.
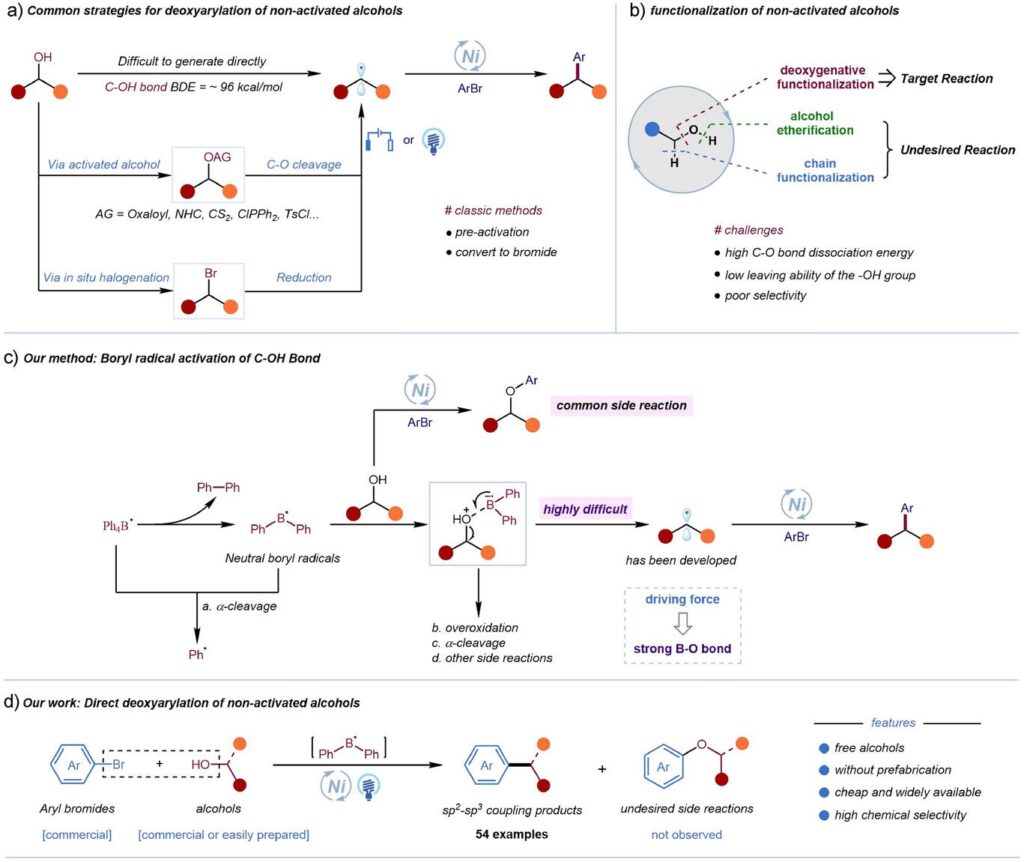
How it works (see the figure):
1) excited 4-CzIPN undergoes single electron transfer with Ph₃N to generate triphenylamine radical cation
2) This radical cation oxidizes tetraphenylboron anion to generate tetraphenylboron radicals, which decompose to form diphenylboron radicals
3) The electron-deficient diphenylboron radical coordinates with the alcohol’s oxygen atom to form a complex intermediate that activates the C-O bond;
4) The resulting alkyl radical is captured by nickel catalyst for reductive elimination.
Reaction conditions
➤ Sodium tetraphenylborate (NaBPh₄) as the boron reagent
➤ Nickel catalyst (Ni(hfacac)₂)
➤ Photocatalyst (4-CzIPN)
➤ 400 nm blue LED irradiation
➤ Triphenylamine (Ph₃N) as an electron donor
➤ 1,2-dichloroethane (DCE) as solvent
➤ Molecular sieves to exclude water
➤ Coupling partners: aryl bromide and primary or secondary alcohol
My highlights
➤ C-C cross coupling is the most prominent type of synthetic transformation in medchem and organic synthesis
➤ Primary and secondary alcohols represent practically unlimited pool of abundant and cheap building blocks.
➤ Mild photoredox conditions with commercially available reagents
➤ 54 substrate examples demonstrate broad scope and good yields
➤ Tolerates diverse functional groups and even complex natural products (stigmasterol, geraniol derivatives, ibuprofen analogs)
I am not a big fan of inflated statements but this methodology might be trully enabling, especially for generation of compound libraries and parallel synthesis.
And another positive fact is that all reagents are commercially available and mostly affordable!
Share your opinion
Let me know your thoughts on the applicability of this procedure!
Are you going to try this protocol in your lab? Let me know how it worked!
Full research paper: https://onlinelibrary.wiley.com/doi/full/10.1002/anie.202520148?saml_referrer