Smart Molecules Targeting Cancer Powerhouse
How can bioorthogonal chemistry and mitochondrial targeting rewrite cancer immunotherapy?
By creating enzyme-responsive nanoparticles that self-assemble in tumors and chemically “click” with an arsenic payload, scientists achieved complete tumor eradication and durable immune memory.
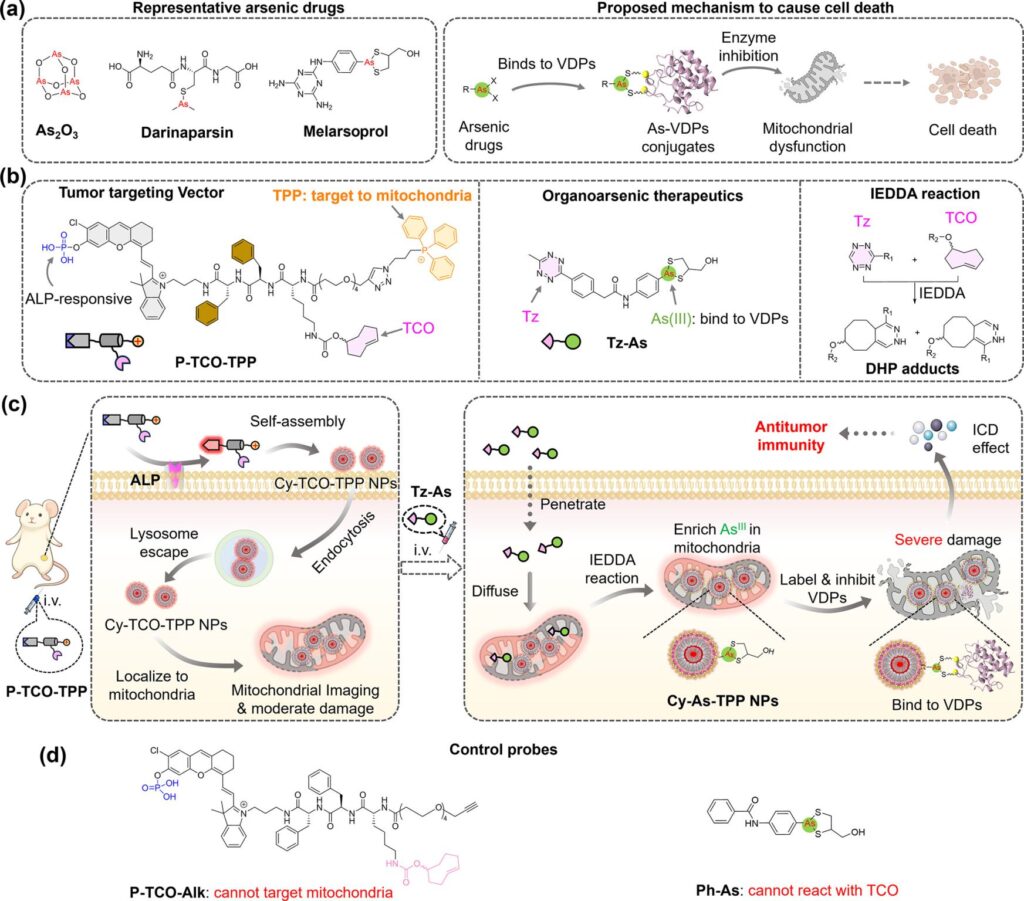
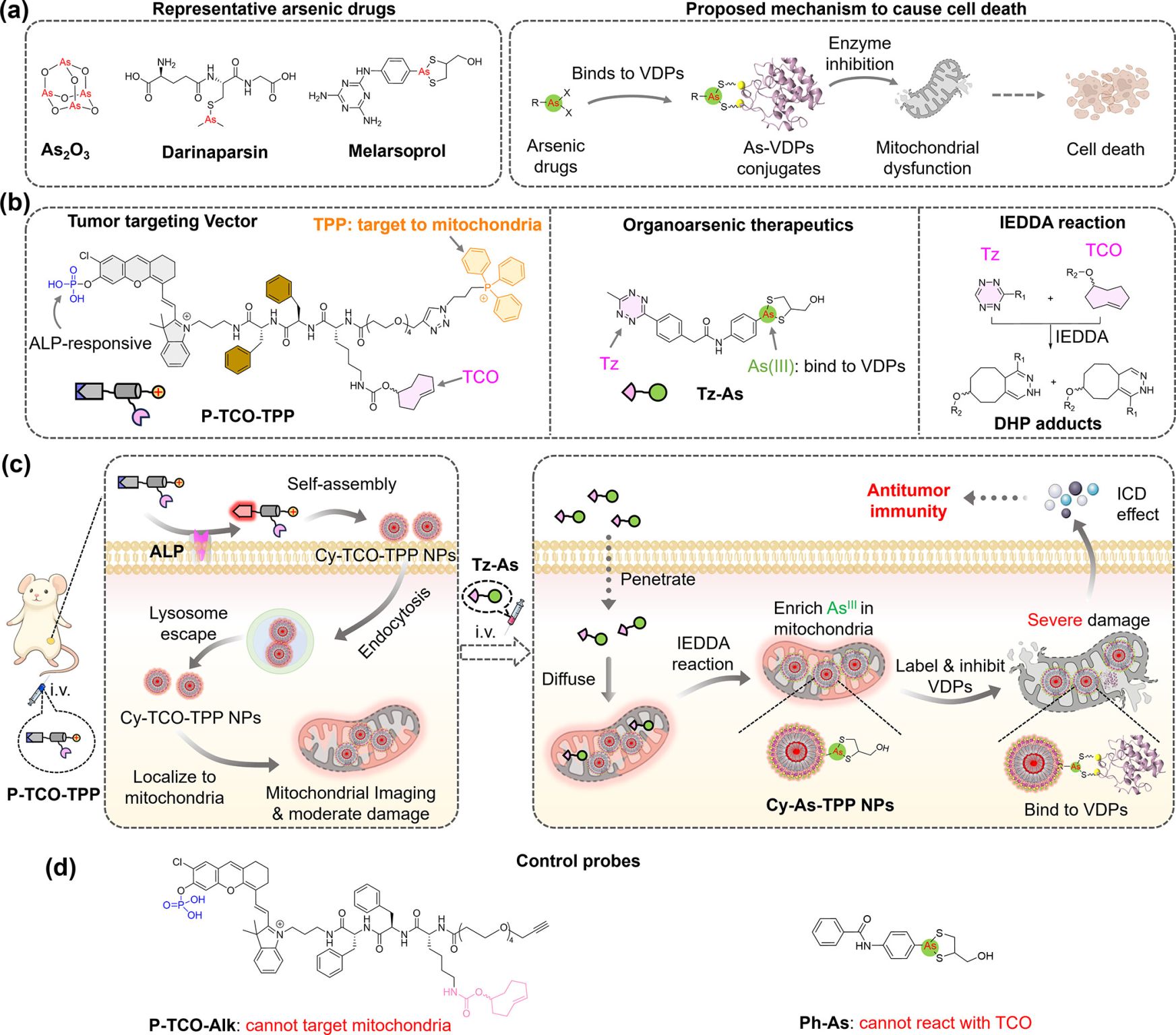
Two main components – two small molecules (Fig. panel b)
💊 𝐏𝐫𝐨𝐛𝐞 𝐏-𝐓𝐂𝐎-𝐓𝐏𝐏:
An ALP-responsive small molecule comprising a phosphate-caged fluorophore (mCy), a D-dipeptide linker (D-FF), a TCO bioorthogonal handle, and a mitochondria-targeting triphenylphosphonium cation.
💊 𝐓𝐡𝐞𝐫𝐚𝐩𝐞𝐮𝐭𝐢𝐜 𝐏𝐚𝐲𝐥𝐨𝐚𝐝 𝐓𝐳-𝐀𝐬:
A tetrazine-As(III)-dithiolane conjugate designed to undergo rapid click reaction with TCO bioorthogonal handle.
How it works (Fig. panel b+c)
1️⃣ Upon systemic administration, P-TCO-TPP penetrates tumors
2️⃣ In ALP-overexpressing tumor cells,
Alkaline phosphatase (ALP) cleaves phosphate from P-TCO-TPP, resulting in Cy-TCO-TPP molecules self-assembly into 80 nm fluorescent nanoparticles
3️⃣ Triphenylphosphonium on the surface of Cy-TCO-TPP nanoparticles promotes endocytosis, escape from lysosomes and mitochondrial localization
4️⃣ Guided by mitochondrial NIR fluorescence, Tz-As is injected at the optimal time to react via IEDDA (inverse demand Diels-Alder reaction) with mitochondrial Cy-TCO-TPP NPs, producing intramitochondrial arsenic nanoparticles.
My highlights
✔️ This two-step strategy effectively overcomes the selectivity and toxicity trade-off of organoarsenical therapies
✔️ Pretargeted treatment exhibits 9.6-fold enhanced cytotoxicity (IC₅₀: 2.75 μM in HeLa; 4.61 μM in 4T1)
✔️ It induces mitochondrial structural damage, ROS generation, caspase-3 activation, and immunogenic cell death
✔️ In mouse models, it halts tumor growth, prevents metastasis, and transforms “cold” tumors into “hot,” enabling 40% complete regression when combined with anti-PD-L1.
Molecules smarter than OpenAI
✅ I like how this approach combines several strategies and mechanisms such as: integrating ALP-triggered release and self-assembly, endosomal transport and mitochondria targeting, fluorescent NIR imaging and bioorthogonal chemistry
✅ Importantly, this approach can be potentially adapted for other payloads and organelles, offering a versatile platform for selective cancer targeting and precision medicine
Let me know what you think!
How do you see pretargeted delivery evolving beyond organoarsenical therapies or beyond oncology?
Could this approach work for other organelles or diseases?
Leave your comment under my LinkedIn post here.
Original research article: https://pubs.acs.org/doi/pdf/10.1021/jacs.5c12201?ref=article_openPDF