From DNA Code to Chemistry
Sumon Pratihar, Eric T. Kool and his team from Stanford University just published new paper in 𝘈𝘯𝘨𝘦𝘸𝘢𝘯𝘥𝘵𝘦 𝘊𝘩𝘦𝘮𝘪𝘦, unlocking a new way of selective RNA modification:
“𝘚𝘦𝘲𝘶𝘦𝘯𝘤𝘦-𝘚𝘱𝘦𝘤𝘪𝘧𝘪𝘤 𝘐𝘯𝘴𝘵𝘢𝘭𝘭𝘢𝘵𝘪𝘰𝘯 𝘰𝘧 𝘈𝘳𝘺𝘭 𝘎𝘳𝘰𝘶𝘱𝘴 𝘪𝘯 𝘙𝘕𝘈 𝘷𝘪𝘢 𝘋𝘕𝘈-𝘊𝘢𝘵𝘢𝘭𝘺𝘴𝘵 𝘊𝘰𝘯𝘫𝘶𝘨𝘢𝘵𝘦𝘴”
Key innovation
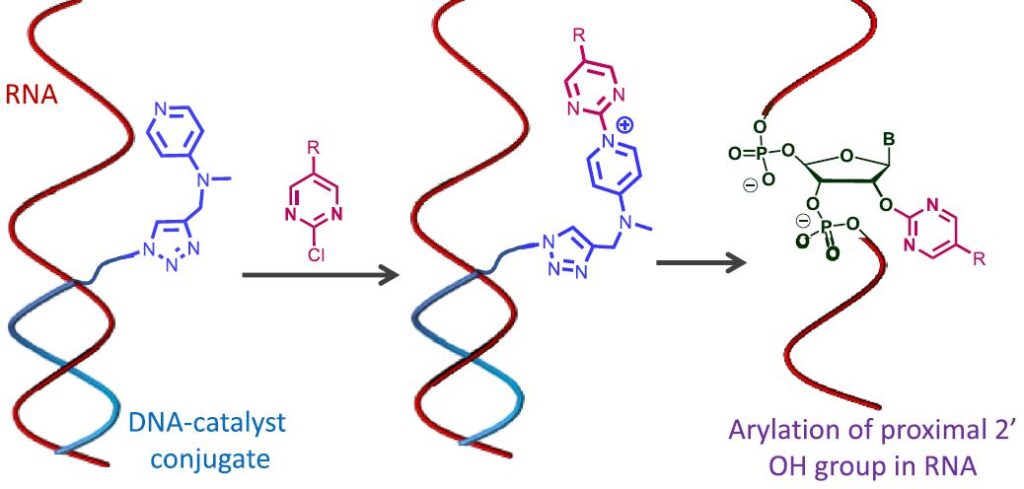
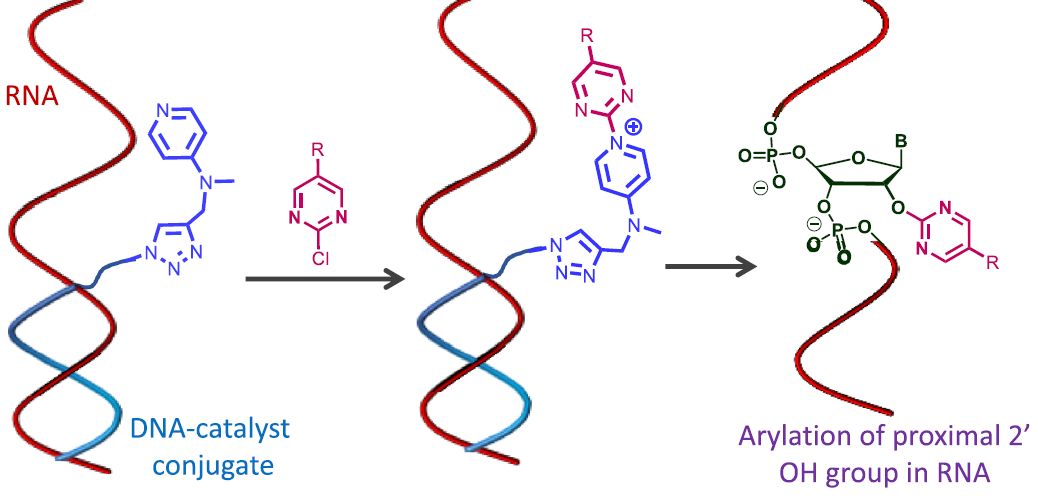
DNA-directed approach that utilizes a DNA oligonucleotide carrying a nucleophilic amine to promote site-selective SNAr reaction at RNA 2′-OH groups proximal to the catalytic group.
How it works
1️⃣ DNA oligonucleotide will recognize and bind to RNA
2️⃣ Chloro-pyrimidine electrophile reacts with DMAP-DNA conjugate (DMAP moiety works as a catalyst)
3️⃣ Transient ammonium aryl intermediate then reacts with RNA near the DNA binding site (in the range of 2-3 nucleotides)
Advantages
✔️ Simpler and more versatile RNA modification than existing methods
✔️ Aryl precursors (tag precursors) are widely available and could be synthetically modified
✔️ This approach does not require custom DNA synthesis nor synthetically modified nucleotides
✔️ Without the necessity of masking the entire RNA
Highlights
✅ The authors selectively arylated a site in the protein-coding region of mCherry mRNA in a mixture of other eGFP and mCherry mRNAs. After transfection of this mixture into HeLa cells, this resulted in 75% knockdown in mCherry protein expression.
✅ This work represents the first report of short complementary DNA-directed site-specific modulation and labeling of messenger RNAs
✅ Unlocks new possibilities for live-cell imaging, mechanistic understanding or therapeutic intervention through site-specific modification of mRNAs
Share your opinion
🔸 What additional chemistry do you envision to be applicable for RNA modification ❓
🔸 How would you deliver the modified DNA oligonucleotide to living cells to enable direct in cellulo applications ❓
Leave your comment under my LinkedIn post here.
Full manuscript: https://doi.org/10.1002/anie.202515681