Breakthrough in Cross‑Coupling Chemistry
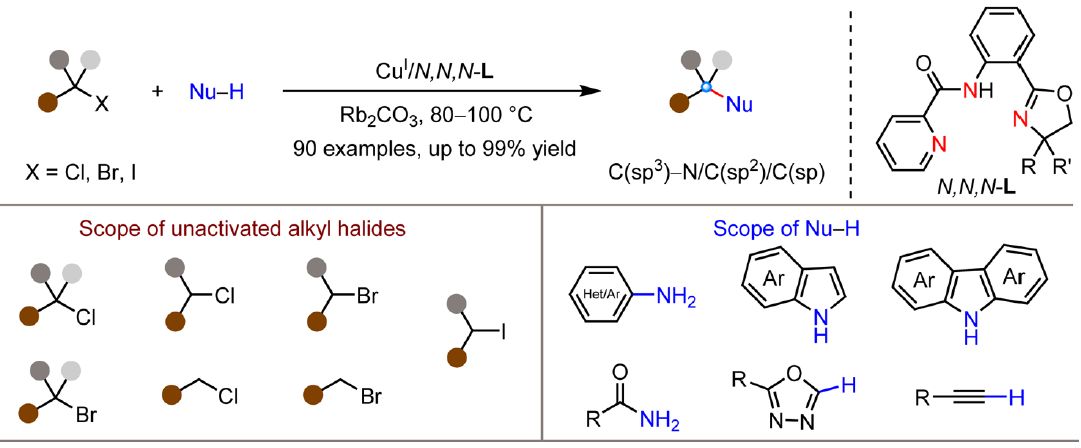
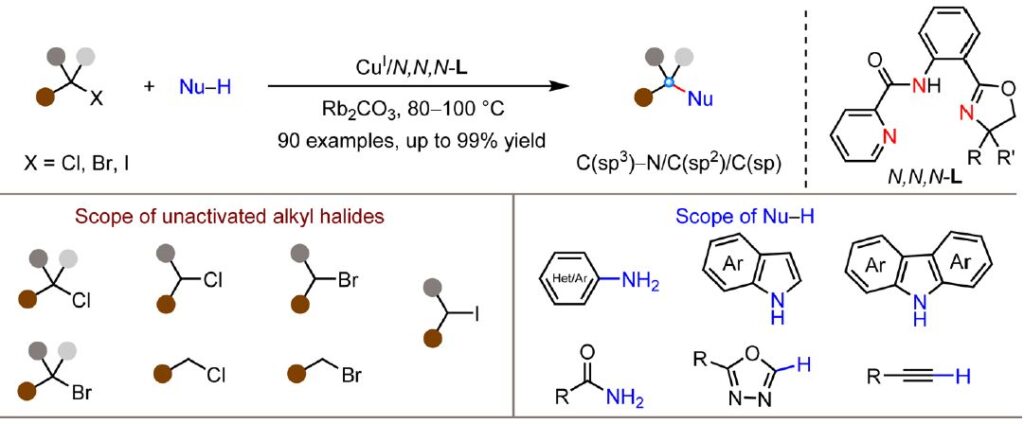
Wang et al. report a new copper-catalyzed methodology for C(sp³)-N cross-coupling of unactivated alkyl halides with diverse nucleophiles under thermal conditions (JACS):
“𝘈 𝘎𝘦𝘯𝘦𝘳𝘢𝘭 𝘊𝘰𝘱𝘱𝘦𝘳-𝘊𝘢𝘵𝘢𝘭𝘺𝘻𝘦𝘥 𝘙𝘢𝘥𝘪𝘤𝘢𝘭 𝘊𝘳𝘰𝘴𝘴-𝘊𝘰𝘶𝘱𝘭𝘪𝘯𝘨 𝘰𝘧 𝘜𝘯𝘢𝘤𝘵𝘪𝘷𝘢𝘵𝘦𝘥 𝘈𝘭𝘬𝘺𝘭 𝘏𝘢𝘭𝘪𝘥𝘦𝘴”
(Colleagues doing automated or high-throughput synthesis will love this)
Key Innovation
The success of this methodology relies on the use of anionic N,N,N-ligands to enhance the reducing capability of Cu(I) catalysts for the reduction of alkyl halides. This represents a departure from previous copper-catalyzed systems that required photoactivation or halogen atom transfer strategies to overcome copper’s inherently weaker reducing ability compared to other first-row transition metals
Substrate Scope
Alkyl halides:
✔️ Primary to tertiary bromides (mostly 80–99% yield, 63-77% for tertiary)
✔️ Secondary and tertiary chlorides (56-83% yields)
✔️ Iodides (high efficiency)
Nucleophiles:
✔️ Anilines (73–96%)
✔️ Indoles and carbazoles (49–95%)
✔️ Azoles (46–95%)
✔️ Terminal alkynes (38–85%)
Function Group Tolerance
Ethers, protected amines, alkenes, alkynes, amides, ketones, esters, and free alcohols are well tolerated
Why This Matters
🎯 First general copper system for unactivated alkyl chloride C-N coupling
🎯 Late-stage functionalization of complex bioactive molecules
🎯 Gram-scale synthesis with >90% yields
🎯 Successfully modified drugs like ibuprofen, indomethacin, and cholesterol derivatives
Additional Insights
Similar to Chan-Lam coupling, also this methodology will likely have problems with heterocyclic substrates (due to Cu coordination), as the published scope is lacking these.
PhCF₃ may limit solubility of polar substrates; however, MeCN or dioxane can be used with slightly reduced yields (see Supporting Information)
🧐 What synthetic challenges are you hoping to see solved next ❓
Full manuscript: https://pubs.acs.org/doi/10.1021/jacs.5c10285
Leave your comments under my LinkedIn post here.
#OrganicChemistry #CopperCatalysis #MedicinalChemistry #ChemicalBiology #CrossCoupling #DrugDiscovery