Spiro Spiro Spirocycles Everywhere.
The number of spirocyclic-N-heterocycles has increased dramatically in the recent years. From 10,000 in 2023 ➡️ 90,000 in 2025 (Substructure search on Reaxys).
Functionalized spirocycles are highly valuable synthetic building blocks, especially in medicinal chemistry. 💊
However, if you want to decorate them a little bit more, you are likely tapping into the chemical space of hard-to-get or expensive compounds (if available at all). And of course, you want to have a pure stereoisomer, right?!
To address this synthetic challenge and demand for spirocyclic building blocks, a team led by Prof. John Hartwig just published in JACS:
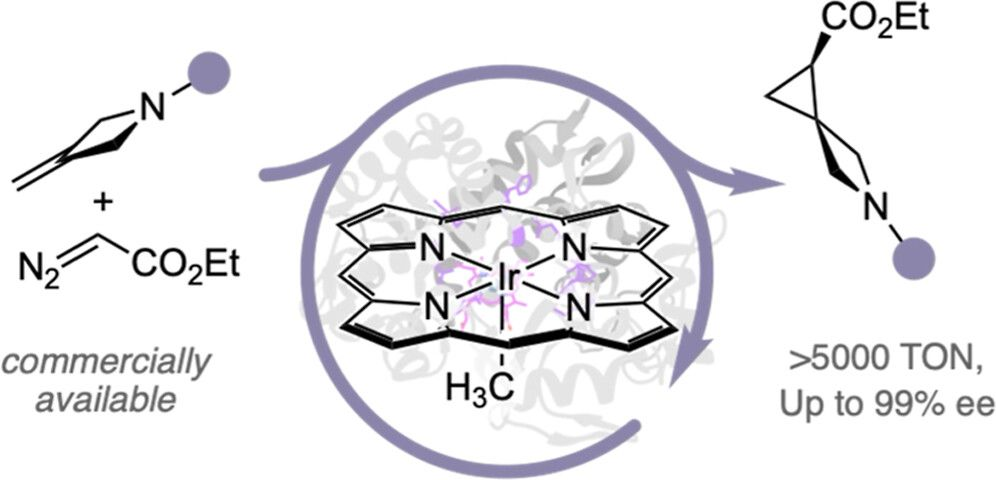
“Enantioselective Synthesis of Spirocyclic Nitrogen-Containing Heterocycles Catalyzed by an Iridium-Containing Cytochrome”
💪 The hero of the story is a metalloenzyme Ir(Me)-CYP119 that catalyzes the asymmetric cyclopropanation of methylene-substituted N-heterocycles.
Key highlights include
✅ Reaction conditions: Metalloenzyme, ethyl diazoacetate (EDA), M9-N buffer, 25 °C, 4 hours.
✅ Ir(Me)-CYP119 has been optimized through directed evolution (4 rounds of 6-codon mutagenesis)
✅ Turnover numbers up to 6,300 TON
✅ Whole-cell catalysis without protein purification at 2 mmol scale possible
✅ Computational investigations have shown that the selectivity is largely determined by the orientation of the carbene intermediate
✅ Rate-determining step is the formation of carbene by extrusion of N₂
A few examples from substrate scope
✅ Methylene azetidines containing:
-Bz and Moc: >95 % ee
-Boc, and Ts: 92 % ee
-Benzoyl substituents with varied steric demand: 65−97 % ee
… leading to corresponding spiroazetidines. (ee = enantiomeric excess)
✅ Authors prepared also spiropyrrolidines and spiropiperidines.
✅ Steric bulk sensitivity varies by ring size and N-substituent.
⚠️ Yields are not discussed in detail in the manuscript. After going through Supporting Information, I can see that yields range from 10 % to 80+%. I am not sure if this is more due to reaction efficiency or purification issues.
Commercial & Process Implications
✅ No exotic reagent, ethyl diazoacetate (EDA) is commercially available
✅ Green, enzymatic alternative to traditional metal catalysts
✅ Array compatibility for high-throughput building-block discovery
✅ Continuous-flow potential for scalable production
✅Opens IP-rich, previously underexplored SpiN chemical space
Significance
✅ Better accessibility of optically pure spirocyclic building blocks will substantially boost the development of traditional small molecules but also new proximity inducing modalities such as PROTACs where optimization of linkers frequently benefits from rigid sp3 rich moieties.
The full manuscript: https://pubs.acs.org/doi/10.1021/jacs.5c06239
Leave your comments under my LinkedIn post here.

hashtag#Spirocycles hashtag#PROTACs hashtag#DrugDiscovery hashtag#Catalysis hashtag#GreenChemistry