Skeletal Editing Strikes Back!
Imagine a scaffold hopping strategy where you don’t have to re-design your synthetic route for each scaffold. You don’t need Jedi tricks nor power of the dark side to achieve that.
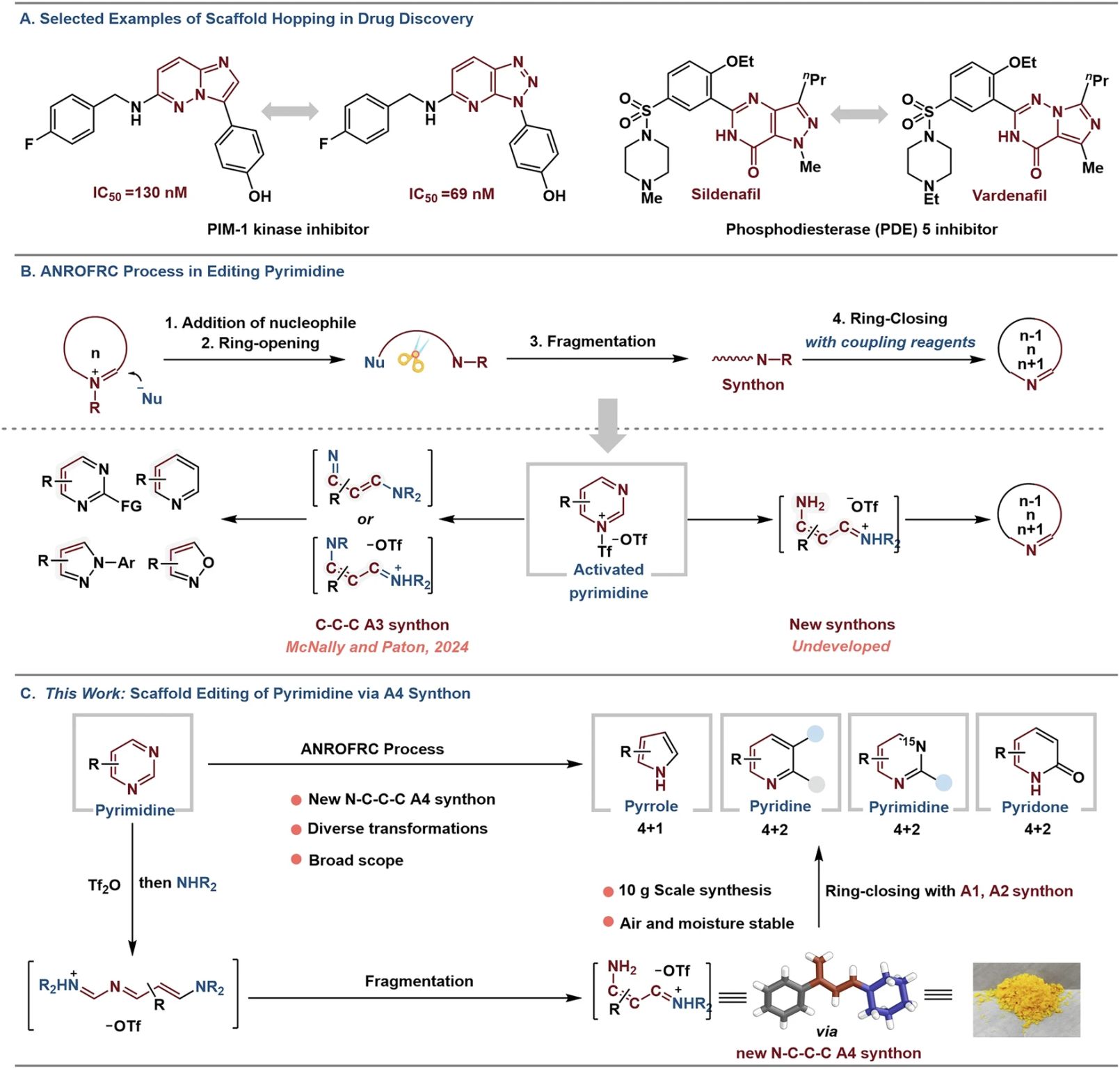
This has been addressed for 4-arylpyrimidines in a recent publication by Shun Li et al. published in Nature Communications:
“Skeletal editing of 4-arylpyrimidines into diverse nitrogen heteroaromatics via four-atom synthons”
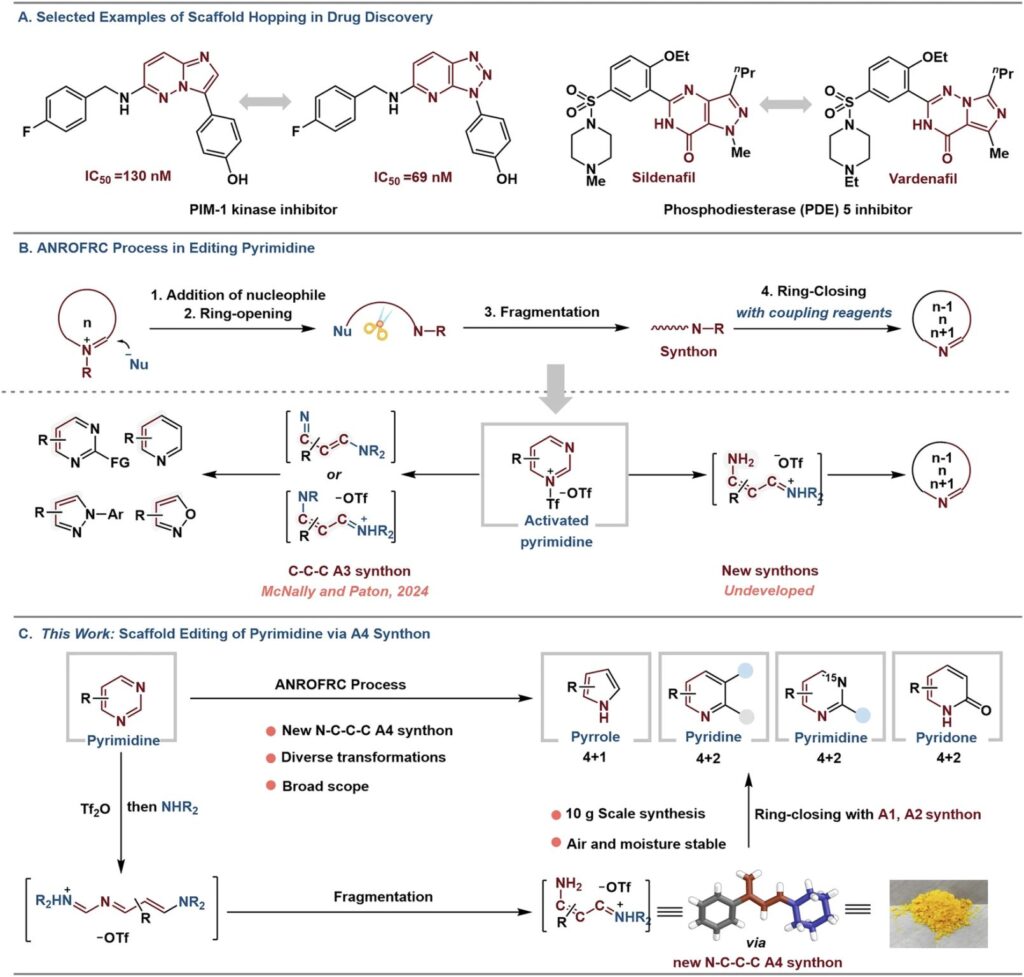
The authors report a “one-to-multiple scaffold-hopping” strategy. They convert 4-arylpyrimidines into two different heterocycles:
✅ Pyrroles by coupling with trimethylsulfoxonium chloride under KOH/THF (4+1 cyclization)
✅ Pyridines by reaction with ketones under KOtBu/1,4-dioxane (4+2 cyclization)
Both transformations proceed through an isolable vinamidinium salt, an N-C-C-C four-atom synthon (A4), obtained in 85–95% yield (X-ray confirmed). This intermediate serves as a unique N-C-C-C four-atom synthon that reacts with a variety of substrates (A1 and A2 synthons), providing substituted pyroles and pyridines.”

Authors further show that the isolated vinamidinium salt can be used for construction of other heterocycles;
✅ Pyridones via cyclization with dialkyl malonates (4+2 cyclization)
✅ ¹⁵N-labeled pyrimidines using ¹⁵N-formamide and KOtBu/DMA
✅ Isoxazoles by using hydroxyl amine
✅ N-Ph-pyrazoles by utilisation of Ph-hydrazine
Key highlights include:
✅ Substrate scope: Over 25 substrates including ketones, aldehydes, nitriles, amides and more give moderate to good yields (40–75%).
✅ Dual synthon behavior: Vinamidinium salt functions as both A4 and A3 synthon (see Fig 1 for structures).
✅ Functional-group tolerance: Electron-rich/poor aryl, heteroaryl and moderately complex motifs converted.
✅ Access to ¹⁵N labeled pyrimidines: Site-specific N1 incorporation for mechanistic or tracer studies.
✅ Practical conditions: Air- and moisture-stable; standard lab setup (light-shielded for pyrroles)
✅ Demonstrated on gram scale (7.0 mmol, 1.09 g) without loss of efficiency.

Main limitations:
⚠️ Strict requirement for C4-aryl substituents (alkyl or unsubstituted C4 failed)
⚠️ Efficiency drop with highly functionalized substrates (e.g., 21% yield with bioactive derivative).
⚠️ Competitive Tf₂O over-triflation and pyrrole light sensitivity.
⚠️ The presence of nucleophilic moieties is going to be problematic.
🧐 Questions for discussion;
What new heterocycles or scaffolds could emerge from utilisation Vinamidinium salt?
Can this be integrated into late-stage functionalization of your small molecules?
⏳Challenge:
Can you rationalize all the steps in the proposed mechanism? (See the scheme below)
Bonus question: What is ANRORC reaction?
Read the full paper: https://lnkd.in/eTEcP-VD
Leave your comment on my LinkedIn here.


#ScaffoldHopping #SkeletalEditing #Heterocycles #DrugDiscovery #VinamidiniumSynthon #MedicinalChemistry #OrganicSynthesis